As previously stated, the aluminum alloys are in a comparatively soft state immediately after quenching from a solution heat treating temperature. To obtain their maximum strengths, they must be either naturally aged or precipitation hardened.
During this hardening and strengthening operation, precipitation of the soluble constituents from the supersaturated solid solution takes place. As precipitation progresses, the strength of the material increases, often by a series of peaks, until a maximum is reached. Further aging (overaging) causes the strength to steadily decline until a somewhat stable condition is obtained. The submicroscopic particles that are precipitated provide the keys or locks within the grain structure and between the grains to resist internal slippage and distortion when a load of any type is applied. In this manner, the strength and hardness of the alloy are increased.
Precipitation hardening produces a great increase in the strength and hardness of the material with corresponding decreases in the ductile properties. The process used to obtain the desired increase in strength is therefore known as aging, or precipitation hardening.
The strengthening of the heat treatable alloys by aging is not due merely to the presence of a precipitate. The strength is due to both the uniform distribution of a finely dispersed submicroscopic precipitate and its effects upon the crystal structure of the alloy.
The aging practices used depend upon many properties other than strength. As a rule, the artificially aged alloys are slightly overaged to increase their resistance to corrosion. This is especially true with the artificially aged high copper content alloys that are susceptible to intergranular corrosion when inadequately aged.
The heat treatable aluminum alloys are subdivided into two classes, those that obtain their full strength at room temperature and those that require artificial aging.
The alloys that obtain their full strength after 4 or 5 days at room temperature are known as natural aging alloys. Precipitation from the supersaturated solid solution starts soon after quenching, with 90 percent of the maximum strength generally being obtained in 24 hours. Alloys 2017 and 2024 are natural aging alloys.
The alloys that require precipitation thermal treatment to develop their full strength are artificially aged alloys. However, these alloys also age a limited amount at room temperature, the rate and extent of the strengthening depending upon the alloys.
Many of the artificially aged alloys reach their maximum natural or room temperature aging strengths after a few days. These can be stocked for fabrication in the -T4 or -T3 temper. High zinc content alloys such as 7075 continue to age appreciably over a long period of time, their mechanical property changes being sufficient to reduce their formability.
The advantage of -W temper formability can be utilized, however, in the same manner as with natural aging alloys; that is, by fabricating shortly after solution heat treatment, or retaining formability by the use of refrigeration.
Refrigeration retards the rate of natural aging. At 32° F, the beginning of the aging process is delayed for several hours, while dry ice (-50° F to -100° F) retards aging for an extended period of time.
Precipitation Practices
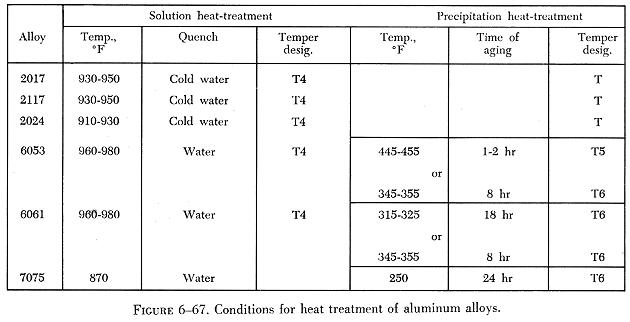
The temperatures used for precipitation hardening depend upon the alloy and the properties desired, ranging from 250° F to 375° F. They should be controlled within a very narrow range (plus or minus 5°) to obtain best results. (See figure 6-67.)

The time at temperature is dependent upon the temperature used, the properties desired, and the alloy. It ranges from 8 to 96 hours. Increasing the aging temperature decreases the soaking period necessary for proper aging. However, a closer control of both time and temperature is necessary when using the higher temperatures.
After receiving the thermal precipitation treatment, the material should be air cooled to room temperature. Water quenching, while not necessary, produces no ill effects. Furnace cooling has a tendency to produce overaging.