CHAPTER 4. CORROSION PRONE AREAS AND PREVENTATIVE MAINTENANCE
|
400. GENERAL.
This chapter lists corrosion problem areas that are common to most aircraft.
Corrosion prone areas should be cleaned, inspected, and treated more frequently
than less corrosion prone areas. The list is not necessarily complete for
any specific aircraft, but could be used to set up a maintenance inspection
program. Also included in this chapter are factors in corrosion control
and preventative maintenance.
401. EXHAUST TRAIL AREAS.
Both jet and reciprocating engine exhaust gas deposits are very corrosive.
Inspection and maintenance of exhaust trail areas should include attention
to the areas indicated in Figure 4-1. Inspection
should also include the removal of fairings and access plates located in
the exhaust gas path. a. Gaps, seams, hinges, and fairings are some of the exhaust trail areas
where deposits may be trapped and not reached by normal cleaning methods.
|
|
b. Exhaust deposit buildup on the upper and lower wing, aft fuselage,
and in the horizontal tail surfaces will be considerably slower and sometimes
completely absent from certain aircraft models.
402. BATTERY COMPARTMENTS AND BATTERY VENT OPENINGS.
In spite of protective paint systems and extensive sealing and venting
provisions, battery compartments continue to be corrosion problem areas.
Fumes from overheated battery electrolyte are difficult to contain and
will spread to internal structure. Unprotected surfaces will be subjected
to corrosive attack. For lead acid batteries, frequent cleaning and neutralization
of acid deposits with sodium bicarbonate solution will minimize corrosion.
If the battery installation includes external vent openings on the aircraft
skin, these areas should be included in the inspection and maintenance
procedure. If aircraft batteries with electrolytes of either sulfuric acid
or potassium hydroxide are in use, their leakage will cause corrosion.
Consult the applicable maintenance manuals for the particular aircraft
to determine which type battery was installed and the recommended maintenance
practices for each. Cleaning of nickel cadmium compartments should be done
with ammonia or boric acid solution, allowed to dry thoroughly, and then
painted with an alkali resistant varnish.
403. LAVATORIES, BUFFETS, AND GALLEYS.
These areas, particularly deck areas behind lavatories, sinks, and ranges,
where spilled food and waste products may collect if not kept clean, are
potential trouble spots. Even if some contaminants are not corrosive in
themselves, they will attract and retain moisture and, in turn, cause corrosive
attack. Pay attention to bilge areas located under galleys and lavatories.
Clean these areas frequently and maintain the protective sealant and paint
finishes. {Fig. 4-2}
404. BILGE AREAS.
On all aircraft, the bilge area is a common trouble spot. The bilge
is a natural sump or collection point for waste hydraulic fluids, water,
dirt, loose fasteners, drill chips, and other odds and ends of debris.
Residual oil quite often masks small quantities of water which settle to
the bottom and set up a hidden potential corrosion cell. With the exception
of water displacing corrosion preventative compounds, keeping bilge areas
free of all extraneous material, including water and oil will insure the
best protection against corrosion. A good vacuum cleaner and clean wiping
cloths are necessary to clean such areas.
405. WHEEL WELLS AND LANDING GEAR.
The wheel well area probably receives more punishment than any other
area of the aircraft. It is exposed to mud, water, salt, gravel, and other
flying debris from runways during flight operations (see Figure
4-3).
a. Frequent cleaning lubrication, and paint touchup are needed on aircraft
wheels and on wheel well areas. Because of the many complicated shapes,
assemblies, and fittings in the area, complete coverage with a protective
paint film is difficult to attain. Thus, preservative coatings tend to
mask trouble rather than prevent it. Because of the heat generated from
braking, preservative coatings cannot be used on aircraft landing gear
wheels.
b. During inspection of this area, particular attention should be given
to the following trouble spots:
(1) High strength steel.
(2) Exposed surfaces of struts, oleos, arms, links, and attaching hardware
(bolts, pins, etc.).
(3) Axle interiors.
(4) Exposed position indicator switches and other electrical equipment.
(5) Crevices between stiffeners, ribs, and lower skin surfaces which
are typical water and debris traps.
(6) Magnesium wheels, particularly around bolt heads, lugs, and wheel
web areas.
(7) Exposed rigid tubing, especially at "B" nuts and ferrules under
clamps and tubing identification tapes.
406. EXTERNAL SKIN AREAS.
External aircraft surfaces are ordinarily covered with protective finishes.
In addition, paint coatings may be applied. The affected external aircraft
surfaces are readily visible or available for inspection and maintenance.
Much emphasis has been given to these areas in the past, and maintenance
procedures are well established. Even here, certain types of configurations
or combinations of materials become troublesome and require special attention
if serious corrosion difficulties are to be avoided. Some of the common
trouble areas, other than those attributed to engine exhaust deposits,
are grouped as follows:
a. Steel, Titanium, CRES, and Nickel Alloy Fasteners.
Fasteners and areas around these fasteners are trouble spots. These
areas are subject to high operational loads, moisture intrusion, and dissimilar
metal skin corrosion (see Figure 4-4).
b. Faying Surfaces and Crevices.
Similar to corrosion around fasteners, corrosion in faying surfaces,
seams, and joints is caused by the intrusion of moisture and other corrosive
agents. The effect of this type of corrosion is usually detectable by bulging
of the skin surface. {See unreferenced figure 4-5.
Crack and skin bulging caused by corrosion.
c. Magnesium Skins.
Properly surface treated, insulated, and painted magnesium skin surfaces
give relatively little trouble from a corrosion standpoint if the original
surface is maintained. However, trimming, drilling, and riveting destroy
some of the original surface treatment which may not be completely restored
by touchup procedures.
(1) Some aircraft have steel fasteners installed through magnesium skin
with only protective finishes under the fastener heads, and fillet sealant
or tape over the surface for insulation. Further, all paint coatings are
inherently thin at abrupt changes in contour, such as at trimmed edges.
With magnesium's sensitivity to moisture, all of these conditions add up
to a potential corrosion problem whenever magnesium is used.
(2) Any inspection for corrosion should include all magnesium skin surfaces,
as well as other magnesium fittings or structural components, with special
attention to edges, areas around skin edges and fasteners, and cracked,
chipped, or missing paint.
d. Spotwelded Skins.
Corrosion of this type construction is chiefly the result of the entrance
and entrapment of moisture or other corrosive agents between layers of
the metal (see Figure 4-6).
(1) Spotwelded assemblies are particularly corrosion prone. Corrosive
attack causes skin buckling or spotweld bulging, and eventually spotweld
fracture. Some of the corrosion may be caused originally by fabricating
processes, but its progress to the point of skin bulging and spotweld fracture
is the direct result of moisture or other corrosive agents working its
way through open gaps or seams. The use of weld through sealing materials
is expected to minimize this problem, but many in-service aircraft still
have unsealed spotweld skin installed. This type of corrosion is evidenced
by corrosion products appearing at the crevices through which the corrosive
agents enter.
(2) Corrosion may appear at either external or internal faying surfaces,
but it is usually more prevalent on external areas. More advanced corrosive
attack causes skin buckling and eventual spotweld fracture. Skin buckling
in this early stage may be detected by sighting or feeling along spotwelded
seams or by using a straight edge.
(3) To prevent this condition, keep potential moisture entry points
including gaps, seams, and holes created by broken welds filled with noncorrosive
sealant.
e. Piano type Hinges.
These are prime spots for corrosion due to dissimilar metal contact
between the steel pin and aluminum hinge tangs. They are also natural traps
for dirt, salt, and moisture. Where this type of hinge is used on access
doors or plates, and actuated only when opened during an inspection, they
tend to corrode and freeze in the closed position between inspections.
When the hinge is inspected, it should be lubricated and actuated through
several cycles to ensure complete penetration of the lubricant (see Figures
4-7 and 4-8).
f. Heavy or Tapered Aluminum Alloy Skin Surfaces.
Heavy or thick sections of most heat treated aluminum alloys are susceptible
to pitting or intergranular corrosion and exfoliation of the metal. When
inspecting external skin surfaces, especially around countersunk fastener
heads, look for white or grey powder deposits or metal exfoliation. This
is usually first evident as small raised areas or bumps under paint film.
(1) Treatment of this corrosive attack includes removal of all corrosion
products, i.e., exfoliated metal is blended and polished not to exceed
the limits set by the aircraft manufacturer. If corrosion products remain
after the limits set by the aircraft manufacture have been reached, contact
the aircraft manufacturer or the Federal Aviation Administration (FAA)
for authorized limits. The treatment is not complete until the restoration
of protective surface finishes is accomplished.
(2) Protect reworked areas with a chemical conversion coating, sealant
primer, and top coat if applicable. Reworked areas should be carefully
watched for any indications of renewed corrosive activity.
g. Organic Composites.
Organic composites used in aircraft can cause different corrosion problems
than those normally associated with all metal structure. Composites such
as graphite/epoxy act as a very noble (cathodic) material, creating the
potential for galvanic corrosion. The galvanic corrosion potential coupled
with different methods of attachment (i.e., adhesive bonding, stepped structures,
locking mechanical fasteners, etc.) lead to multicomponent galvanic couples
with the problem being particularly aggravated by high humidity and salt
water environments. Application of aircraft sealants over the dissimilar
metal/composite junctions will prevent galvanic corrosion if moisture is
completely excluded. However, since complete exclusion of moisture is virtually
impossible under extended periods of flight operation, the most effective
method of eliminating the voltage potential is to provide a nonconductive
layer such as fiberglass/epoxy and/or sealant between the composite and
dissimilar metal surfaces.
407. WATER ENTRAPMENT AREAS.
Corrosion will result from the entrapment of moisture. With the exception
of sandwich structures, design specifications usually require that the
aircraft have low point drains installed in all areas where moisture and
other fluids can collect. In many cases, these drains are ineffective either
due to location or because they are plugged by sealants, extraneous fasteners,
dirt, grease, and debris. Potential entrapment areas are not a problem
when properly located drains are functioning, and the aircraft is maintained
in a normal ground attitude. However, the plugging of a single drain hole
or the altering of the level of the aircraft can result in a corrosion
problem if water becomes entrapped in one of these "bathtub" areas. Daily
inspection of low point drains is a recommended practice.
408. ENGINE FRONTAL AREAS AND COOLING AIR VENTS.
Constant abrasion by airborne dirt and dust, bits of gravel from runways,
and rain tends to remove the protective surfaces from these areas. Furthermore,
cores of radiator coolers, reciprocating engine cylinder fins, etc., due
to the requirement for heat dissipation, may not be painted. Engine accessory
mounting bases usually have small areas of unpainted magnesium or aluminum
on the machined mounting surfaces. With moist and salt or industrial pollutant
laden air constantly flowing over these surfaces, they are prime sources
of corrosive attack. Inspection of such areas should include all sections
in the cooling air path with special attention to obstructions and crevices
where salt deposits may build up during marine operations (see Figures
4-9 and 4-10).
409. ELECTRONIC PACKAGE COMPARTMENTS.
Electronic and electrical package compartments cooled by ram air or
compressor bleed air are subjected to the same conditions common to engine
and accessory cooling vents and engine frontal areas. While the degree
of exposure is less because of a lower volume of air passing through and
special design features incorporated to prevent water formation in enclosed
spaces, this is still a trouble area that requires special attention.
a. Circuit breakers, contact points, and switches are extremely sensitive
to moisture and corrosive attack and should be inspected for these conditions
as thoroughly as design permits. If design features hinder examination
of these items while in the installed condition, inspection should be accomplished
after component removal for other reasons.
b. Treatment of corrosion in electrical and electronic components should
be done only by, or under the direction of, qualified personnel familiar
with the function of the unit involved as conventional corrosion treatment
may be detrimental to some units.
c. Most corrosion that occurs on avionic equipment is similar to that
which occurs on the basic airframe structure. The difference between avionic
and airframe corrosion is that minute amounts of corrosion in avionic equipment
can cause serious degradation or complete failure, while it would be unnoticed
on larger structures.
d. Smog, smoke, soot, and other airborne contaminants are extremely
corrosive to exposed avionic equipment. Many fumes and vapors emitted from
factories or industrial complexes are highly acidic and greatly accelerate
corrosion. An example is the corrosive effect of ozone, a product of many
welding machines and large electrical motors. Complete degradation of rubber
seals and damage to delicate components have occurred in equipment stored
near ozone producing equipment. Avionic shops and storage areas should
have a filtered air conditioning system.
e. Another manmade atmosphere is the aircraft environmental control
system. These systems induce cooling air to the equipment. They may include
a filter system that extracts moisture, and in some cases contaminants,
from the air that enters the equipment. Failure to replace and/or clean
a filter, or eliminate a leaky environmental seal, may cause a moisture
or contaminant buildup that could cause a corrosive atmosphere within the
equipment.
f. The control of corrosion in avionic systems is similar to that in
airframes, with appropriate modifications. The general differences in construction
and procedures between airframe and avionics relative to corrosion control
are:
(1) Less durable protection systems;
(2) Small amounts of corrosion can make equipment inoperative;
(3) Dissimilar metals are often in electrical contact;
(4) Stray currents can cause corrosion;
(5) Active metals and dissimilar metals in contact are often unprotected;
(6) Closed boxes can produce condensation during normal temperature
changes during flight;
(7) Avionic systems have many areas to trap moisture;
(8) Hidden corrosion is difficult to detect in many avionic systems;
(9) Many materials used in avionic systems are subject to attack by
bacteria and fungi; and
(10) Organic materials are often used which, when overheated or improperly
or incompletely cured, can produce vapors which are corrosive to electronic
components and damaging to coatings and insulators.
410. MISCELLANEOUS TROUBLE AREAS.
A variety of additional trouble spots exists, and some are covered by
manufacturers' publications. Most aviation activities can add a favorite
to the following list:
a. Examine all flexible hose assemblies for chafing, weather checking,
hardening, discoloration, evidence of fungus, and torn weather protective
coatings or sleeves. Replace those hoses that are found to be discrepant.
b. Trimmed edges of sandwich panels and drilled holes should have some
type of corrosion protection. A brush treatment with an inhibitor solution
or the application of a sealant along the edge, or both, is recommended.
Any gaps or cavities where moisture, dirt, or other foreign material can
be trapped should be filled with a sealant. The adjacent structure (not
the sandwich) should have sufficient drainage to prevent moisture accumulation.
Damage or punctures in panels should be sealed as soon as possible to prevent
additional moisture entry - even if permanent repair has to be delayed.
c. Control cables may present a corrosion problem whether carbon steel
or stainless steel is used. The presence of bare spots in the preservative
coating is one of the main contributing factors in cable corrosion. Cable
condition should be determined by cleaning the cable assembly, inspection
for corrosion, and application of an approved preservative if no corrosion
is found. If external corrosion is found, relieve tension on the cable
and check internal strands for corrosion. Cables with corrosion on internal
strands should be replaced. Pay particular attention to sections passing
through fairleads, around sheaves, and grooved bellcrank arms. External
corrosion should be removed by a clean, dry, coarse rag or fiber brush.
After complete corrosion removal, apply a preservative.
d. Topcoating materials (Buna - N, Polyurethane, and Epoxy) used in
integral fuel cells are impervious to fuel but not completely impervious
to moisture absorption. Since it is impossible to keep fuel completely
free of water, moisture can penetrate through the topcoating materials
and sometimes causes pitting or intergranular corrosion on aircraft structural
parts. It has also been found that micro-organisms which live in the water
entrained by fuel, particularly jet propellant types, feed on fuel hydrocarbon
and hydrocarbon type elastomeric coatings materials. These micro-organisms
excrete organic acids, and dead micro-organisms act as a gelatinous acidified
sponge which can deteriorate integral tank coatings and corrode the aircraft
structure. Microbial corrosion can be minimized by preventing as much water
contamination of the fuel as possible with well managed storage facilities,
adequate filtration of fuel, and drainage of water contamination from integral
fuel cells which keeps the water moving and reduces the chance for the
colonies of microorganisms to develop. Micro-organic activity can be reduced
by using a biocide additive such as "Biobor JF" or equivalent. Solution
strength and application frequency should be in accordance with the manufacturer's
instructions.
e. Electrical connectors/components may be potted with a sealing compound
to provide more reliability of equipment. The sealing compound prevents
entrance of moisture into the area of connectors where the wires are attached
to the pins.
(1) Rubber O-rings may also be used to seal moisture out of the mating
area of pin connections and to prevent loss of pressurization in compartments
containing bulkhead connectors.
(2) Moisture intrusion into electrical connectors can cause corrosion
and an electrical failure. Suspected plugs should be disconnected, dissembled,
solvent cleaned, and inspected for corrosion.
(3) When sealing provisions are not designed into the electrical component,
these components can have moisture intrusion and internal corrosion.
f. Severe corrosion damage to the rear pressure bulkhead below the floor
may occur as a result of contamination by fluids. Inspection for rear bulkhead
corrosion may require extensive disassembly of components and fixtures
to allow a thorough visual inspection. When inspection access holes are
available, inspection by fiber optics is useful. Other nondestructive inspection
(NDI) methods (X-ray, ultrasonic, and eddy current) are also available.
However, these inspection techniques require specially trained personnel,
NDI comparison standards, and suitable access. A regular inspection of
the rear pressure bulkhead (both front and rear faces) below the floor
level should be accomplished to prevent serious corrosion from occurring
between the bulkhead and periphery doubler at the floor level. Such corrosion
could weaken the bulkhead skin and cause sudden cabin pressure loss.
g. Some older aircraft have developed delaminations in cold bonded joints.
Corrosion between the delaminated surfaces is caused by moisture intrusion
along the edge of the mating parts or around fasteners securing the mating
parts together. Localized bulging of the skin or internal structural component,
usually around the fasteners, is the first indication of a corrosion problem
(see Figure 4-11). Skin cracks or dished or missing
fastener heads may also indicate severe corrosion in bonded joints. Corrosion
which occurs between skins, doublers, and stringers or frames will produce
local bulging or pulled rivets (see Figure 4-12).
Corrosion that occurs between the skins and doublers or tear straps away
from backup structure such as stringer or frame will not produce local
bulging. An external low frequency eddy current inspection may be used
to determine the extent of corrosion in the skin. Lap joints should be
opened with wedges to determine the full extent of corrosion damage (see
Figures 4-13 and 4-14). Internal
visual inspection should be used to detect delaminated doublers or tear
straps. A penetrating water displacement corrosion inhibitor should be
applied to faying surfaces after corrosion removal and repair.
h. Flap and slat recesses and equipment installed in these areas, which
are normally closed, may corrode unnoticed unless special inspections are
performed.
411. FACTORS IN CORROSION CONTROL.
a. Corrosion Factors.
The degree of severity, the cause, and the type of corrosion depend
on many factors, including the size or thickness of the part, the material,
heat treatment of the material, protective finishes, environmental conditions,
preventative measures, and design.
(1) Thick structural sections are generally more susceptible to corrosive
attack because of variations in their composition, particularly if the
sections are heat treated during fabrication. When large sections are machined
or chem-milled after heat treatment, the corrosion characteristics of thinner
sections may be different from those of thicker areas. Section size is
based on structural requirements and cannot be changed for the purpose
of controlling corrosion. From a maintenance standpoint, the correct approach
is one of recognizing the need to ensure the integrity and strength of
major structural parts and maintaining permanent protection over such areas
at all times.
(2) In service stresses and field repairs may affect the rates and types
of corrosion. Aircraft structure under high cyclic stresses, such as helicopter
main rotors, are particularly subject to stress corrosion cracking. Also
areas adjacent to weld repaired items often have corrosion due to insufficient
removal of the weld flux, or, for some steels, buildup of a magnetic field.
Areas such as these should be closely inspected for signs of corrosion
and, when found, proper treatment accomplished.
b. Corrosion Control in Design.
Since corrosion is the deterioration of metals resulting from reactions
between metals and their environment, some corrosion control or means to
minimize corrosion when the aircraft enters operational service should
be introduced during the design phase. The corrosion issues discussed in
this AC provide information to reduce the rate of corrosive attack by corrosion
control measures introduced early in design.
(1) The nature of the material is a fundamental factor in corrosion.
High strength, heat treatable aluminum and magnesium alloys are very susceptible
to corrosion, while titanium and some stainless steel alloys are less susceptible
in atmospheric environment. The aircraft manufacturer selects material
for the aircraft based on material strength, weight, and cost, while corrosion
resistance is often a secondary consideration. However, corrosion control
should be considered as early as possible during the preliminary design
phase.
(2) The use of more corrosion resistant materials in any design normally
involves additional weight to achieve required strength. Since weight consideration
is a major factor in the construction of airframes, the primary means of
preventing corrosion is by use of protective coatings and proper maintenance
procedures.
(3) The use of corrosion resistant alloys is not a cure all for corrosion
prevention. A common mistake is to replace a corroded part with a corrosion
resistant alloy only to find that the corrosion has now shifted to another
part and increased in severity.
(4) The problem of protection against corrosion is minimized if the
material to be protected is intrinsically resistant to corrosion. Aluminum
copper alloys are known to have better stress corrosion resistance and
better fatigue strength properties than aluminum zinc alloys; therefore,
they are often used as the primary structural materials.
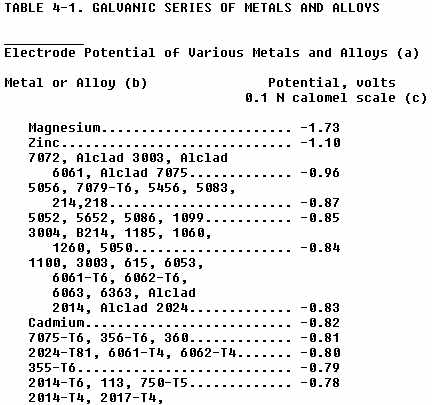
(5) Galvanic corrosion is created by dissimilar metals being in contact
with each other. The galvanic series of metals and alloys (Table 4-1) is
a factor that should be considered in the repair of aircraft. The further
apart the metals listed in Table 4-1 are, the greater the tendency will
be for galvanic corrosion. The metals grouped together in Table 4-2 have
little differences in electrical potential; thus they are relatively safe
to use in contact with one another. However, the coupling of metals from
different groups will result in corrosion of the group having a lower number.
(a) Data from Alcoa Research Laboratories.
(b) The potential of all tempers is the same unless temper is designated.
(c) Measured in an aqueous solution of 53 g of sodium chloride +
3 g hydrogen peroxide per liter at 25 degree C.
(d) The potential varies with quenching rate.
__________
TABLE 4-2. GROUPING OF METALS AND ALLOYS
GROUP I Magnesium and Magnesium Alloys.
GROUP II Aluminum, Aluminum Alloys, Zinc, Cadmium and Cadmium-Titanium
Plate.
Group III Iron, Steels - Except Stainless Steels; Lead, Tin and
their Alloys.
GROUP IV Copper, Brass, Bronze, Copper-Berylium, Copper-Nickel Chromium,
Nickel, Nickel Base Alloys, Cobalt Base Alloys, Carbon Graphite, Stainless
Steels, Titanium and Titanium Alloys.
NOTE:
1. Metals listed in the same group are considered similar to one
another.
2. Metals listed in different groups are considered dissimilar to
one another.
__________
c. Protective Finishes.
Protective finishes provide protection for the base material from corrosion
and other forms of deterioration. Protective finishes are divided into
2 separate categories, sacrificial and nonsacrificial. Sacrificial coatings
include cadmium, zinc, and aluminum. Nonsacrificial coatings include hard
plating (chromium and nickel), chemical conversion coatings, sealant, primers,
and top coat.
d. Geographical Location and Environment.
This factor concerns systems exposed to marine atmospheres, moisture,
acid rain, tropical temperature conditions, industrial chemicals, and soils
and dust in the atmosphere. Limit, whenever possible, the requirement for
operation of aircraft in adverse environments.
(1) Moisture is present in the air as a gas (water vapor) or as finely
divided droplets of liquid (mist or fog) and often contains contaminants
such as chlorides, sulfates, and nitrates, which increase its corrosive
effects. Condensed moisture which evaporates will leave its contaminants
behind. Condensed moisture and its contaminants can also be trapped in
close fitting, wettable joints, such as faying surfaces and be drawn along
poor bond lines by capillary action.
(2) Salt particles, when dissolved in water, form strong electrolytes.
Normal sea winds carry dissolved salt which makes coastal environments
highly corrosive.
(3) Industrial pollutants (such as carbon, nitrates, ozone, sulfur dioxide,
and sulfates) contribute to the deterioration of nonmetallic materials
and can cause severe corrosion of metals.
(4) Warm, moist air, normally found in tropical climates accelerates
corrosion while cold, dry air normally found in arctic climates reduces
corrosion.
e. Heat Treatment.
Proper heat treatment of materials is a vital factor in maximizing resistance
to corrosion.
412. PREVENTATIVE MAINTENANCE.
a. Prevention.
Corrosion prevention of aircraft structure depends on a comprehensive
corrosion prevention and control plan, implemented from the start of operation
of an aircraft, which includes:
(1) Adequately trained personnel in:
(i) Recognition of corrosion inducing conditions;
(ii) Corrosion identification techniques;
(iii) Corrosion detection, cleaning, and treating; and
(iv) Lubrication and preservation of aircraft structure and components.
(2) Inspection for corrosion on a scheduled basis.
(3) Thorough cleaning, inspection, lubrication, and preservation at
prescribed intervals. Suggested intervals based on operating environment
(see Figures 4-15 through 4-20)
are:
{Figs. 4-16, Fig 4-17,
Fig 4-18, Fig 4-19}
(i) Mild zones - Every 90 days;
(ii) Moderate zones - Every 45 days; and
(iii) Severe zones - Every 15 days.
(4) Prompt corrosion treatment after detection.
(5) Accurate record keeping and reporting of material or design deficiencies
to the manufacturer and the FAA.
(6) Use of appropriate materials, equipment, and technical publications.
(7) Maintenance of the basic finish systems.
(8) Keeping drain holes and passages open and functional. Sealants,
leveling compounds, miscellaneous debris, or corrosion inhibitors should
not block drain paths (see Figure 4-21).
(9) Replacing deteriorated or damaged gaskets and sealants (using noncorrosive
type sealants) to avoid water intrusion and entrapment which leads to corrosion.
(10) Minimizing the exposure of aircraft to adverse environments, such
as hangaring away from salt spray.
b. Cleaning Compounds.
NOTE: "More is not always better." Mixing more cleaning compound increases
the pH of the solution which can do more harm than good. A cleaning compound
should always be mixed in accordance with the manufacturer's recommendations.
(1) Cleaning compounds work by dissolving soluble soils, emulsifying
oily soils, and suspending solid soils. There are several types of cleaning
compounds, each of which cleans a surface using one or more of these mechanisms.
(2) Highly alkaline cleaning compounds (pH greater than 10) are not
recommended. Moderately alkaline cleaners (pH between 7.5 and 10) conforming
to MIL-C-85570, Types I and II, are recommended. Both types contain detergents,
foaming agents, and solvents, and work in the same way as a detergent solution.
(3) High gloss spot cleaner conforming to MIL-C-85570, Type III, is
recommended for cleaning exhaust track areas of high gloss paint systems.
This material contains solvents, detergents, and suspended abrasive matter
to remove soil by wearing away the surface that holds it.
(4) Thixotropic (viscous) cleaner conforming to MIL-C-85570, Type V,
is recommended for cleaning wheel wells and replacement of some solvent
cleaning where water rinsing can be tolerated. This cleaner contains solvents,
detergents and some thickening agents. When applied undiluted to an oily
or greasy surface, the cleaner clings long enough to emulsify the soil
(about 5 to 15 minutes) and can then be rinsed away with fresh water.
NOTE: The use of solvents for cleaning operations is becoming more and
more limited due to environmental regulations. Determine local requirements
for waste disposal.
(5) Solvent emulsion cleaners conforming to MIL-C-43616 and MIL-C-85704
become emulsions when diluted. The solvent contained in the cleaner softens
oily soils so that they can be emulsified by the detergent and rinsed away.
(6) Detergent solution cleaners conforming to MIL-D-16791 dissolve in
water and clean by dissolving soluble salts, emulsifying low viscosity
oils, and suspending easily removed dirt and dust. They are not very effective
on grease, but are excellent cleaners for interior lightly soiled areas,
plastics, and instrument glass covers.
(7) Cleaning solvents dissolve oily and greasy soils so that they can
be easily wiped away or absorbed on a cloth. Solvents differ significantly
in cleaning ability, toxicity, evaporation rate, effect on paint, and flammability.
The use of cleaning solvents is intended for localized spot application
only. A dry cleaning solvent conforming to P-D-680, Type II, is the most
common cleaning solvent used on aircraft, due to its low toxicity, minimal
effect on paint, and relative safety. Other solvents, such as alcohols,
ketones, chlorinated solvents, and naphtha, are specialized materials and
have restricted use. Refer to the manufacturer's maintenance and cleaning
procedures for specific applications.
(8) Miscellaneous cleaning agents include:
(i) Plastic polish which contains mild abrasive matter to polish out
scratches in canopy materials;
(ii) Alkaline chemicals used to neutralize specific acidic soils;
(iii) Sodium bicarbonate for electrolyte spills from sulfuric acid
batteries; and
(iv) Monobasic sodium phosphate and boric acid for electrolyte spills
from nickel-cadmium batteries.
(9) Steam cleaning is not recommended for general use on aircraft. It
erodes paint, crazes plastic, debonds adhesives, damages electrical insulation,
and drives lubrication out of bearings.
c. Cleaning Procedures.
(1) The following cleaning procedures are recommended:
(i) Remove/disconnect all electrical power;
(ii) Ground aircraft;
(iii) Aircraft wash personnel should wear protective gear (gloves,
goggles, aprons, etc.);
(iv) Protect against water/cleaning compound intrusion (close doors,
openings, cover vents, pitot static openings, cover wheels, etc.);
(v) Accomplish prewash lubrication. Lubricate in accordance with applicable
maintenance manual;
(vi) Mix cleaning solution to manufacturer's recommendation;
(vii) Use spray not a stream of water during aircraft wash;
(viii) Do not use abrasive cleaning pads; and
(ix) Rinse aircraft with fresh water to remove all cleaning compounds.
(2) The following postcleaning procedures are recommended:
(i) Remove all covers, plugs and masking materials;
(ii) Inspect and clear all drain holes;
(iii) Inspect open and all known water trap areas for water accumulation
and proper drainage;
(iv) Lubricate aircraft in accordance with applicable maintenance manual;
and
(v) Apply operational preservation.
d. Preservation.
The day to day application of corrosion preventive compounds is used
to protect metal aircraft parts and components. They function by preventing
corrosive materials from contacting and corroding bare metal surfaces.
Many of these compounds are also able to displace water and other contaminants
from the surfaces to be protected. Some also provide lubrication as well
as corrosion protection. Corrosion preventive compounds vary in appearance
and consistency from thick, black types to light oils. Some are water displacing
and others are not. The thicker compounds provide the best corrosion protection,
are longer lasting, and are more difficult to remove. The thinner materials
provide some lubrication and do not crack, chip, or peel but must be removed
and replaced regularly to provide continuing protection.
e. Surface Treatment.
(1) An important step in the corrosion control process is the surface
treatment of the metal with a prescribed chemical to form a protective
film. Chemical surface treatments properly applied provide corrosion resistance
to the metal and improve the adhesion of subsequently applied paints. These
surface treatments, also known as chemical conversion coatings, chromate
conversion coatings, chemical films, or pretreatments, are aqueous acid
solutions of active inorganic compounds which convert aluminum or magnesium
surfaces to a corrosion resistant film.
(i) Aluminum and aluminum alloys chemical conversion materials conform
to MIL-C-81706.
(ii) Magnesium alloy chemical conversion materials conform to MIL-M-3171.
(iii) Ferrous metals, stainless steel, and titanium treatment prior
to painting are limited to corrosion removal and cleaning.
(2) The surface should be prepared for application of the chemical conversion
coatings.
(i) Feather the edges of paint along the edge of areas that have been
chemically stripped prior to pretreatment and repainting to ensure a smooth,
overlapping transition between the old and new paint surfaces.
(ii) Clean the area with a fine or very fine clean abrasive mat saturated
with water.
(iii) Rinse by flushing with fresh water. Particular attention should
be given to fasteners and other areas where residues may become entrapped.
At this stage in the cleaning, the surface should be water break free.
A surface showing water breaks (water beading or incomplete wetting) is
usually contaminated with grease or oil, which will later interfere with
conversion coating, sealing, and painting.
(iv) If the surface is not free of water breaks, reclean the area with
a solution of aircraft cleaning compound using the abrasive mat. Rinse
thoroughly with water.
(3) Chemical conversion coating is applied by brush, sponge stick moistener,
or nonatomizing sprayer to achieve a yellow to gold color for aluminum
(usually 2 to 4 minutes) or greenish brown, brassy or brownish-yellow for
magnesium (30 seconds to 2 minutes).
(4) Immediately rinse the part thoroughly with fresh water. Do not wipe
with a rag or cloth.
(5) Allow the chemical conversion coated surface to dry a minimum of
1/2 hour and a maximum of 4 hours before painting.
f. Sealants.
(1) Sealants are one of the most important tools for corrosion prevention
and control. They prevent the intrusion of moisture, salt, dust, and aircraft
fluids, which can lead to extensive corrosion. For sealants to be effective,
it is critical that the correct sealant be chosen for a specific area/situation
and that it be applied correctly.
(2) Sealants are used for the following reasons:
(i) Fuel tank sealing;
(ii) Pressure areas;
(iii) Weather sealing;
(iv) Firewalls;
(v) Electrical;
(vi) Acid resistant areas;
(vii) Windows;
(viii) High temperature applications; and
(ix) Aerodynamic sealing.
(3) There are numerous sealing compounds available with different properties
and intended use. Refer to the aircraft manufacturer's manual for specific
information concerning selection of the sealing compound and proper application.
Observe the warning and cautions of the manufacturer when using sealing
compounds. Sealing compounds generally are divided into two major types,
those requiring a curing agent and those which cure in air.
(i) Polysulfide, polythioether, and polyurethane sealing compounds consist
of the base (prepolymer) and the accelerator (curing agent). When thoroughly
mixed, the catalyst cures the base to a rubbery solid. Rates of cure depend
on the type of base, catalyst, temperature, and humidity. A full cure may
not be achieved for as long as 7 days.
(ii) Silicone sealing compounds generally consist of one component which
cures by reaction with moisture in the air. If silicones are applied too
thick or in such a way as to prevent moisture from entering the material,
they may not cure at all. In addition, many silicone sealing compounds
produce acetic acid (vinegar smell) while curing, which can lead to severe
corrosion problems. The use of silicone sealing compounds on aircraft should
be limited to those noncorrosive products conforming to Specification (MIL-A-46146).
(iii) Some sealing compounds may require the application of a special
primer or adhesion promoter prior to sealant application in order to develop
a good adhesive bond with the surface. Use only those primers or adhesion
promoters recommended by the product manufacturer.
(4) Sealant application procedures:
(i) Following the removal of corrosion and application of a chemical
surface treatment, prime all surfaces, except internal fuel tank surfaces.
If the surfaces have been contaminated following surface treatment, clean
the area with cleaning solvent and a clean cloth. Dry the surfaces immediately
with a clean cloth. Do not allow solvent to evaporate from the surface.
(ii) Mask off the area being sealed to prevent sealant from contacting
adjacent areas during application and postapplication smooth out operations.
Examples of where masking may be beneficial are fillet sealing of exterior
surface lap and butt seams.
(iii) When required by the manufacturer, apply a thin coating of an
adhesion promoting solution. Allow to dry by evaporation without touching
the area for 30 minutes to 1 hour before applying sealant.
(iv) Spatula type sealants may be applied with a nonmetallic spatula
or scraper. Avoid the entrapment of air. Work sealant into recesses by
sliding the edge of the scraper firmly back over the recesses between the
tape. Smoothing will be easier if the nonmetallic scraper is first dipped
in water.
(v) Sealant to be applied with a brush is applied and smoothed until
the desired thickness is reached.
(vi) Sealant to be applied with a caulking gun will not usually require
masking and is especially adaptable to filling seams or the application
of form in place gaskets.
(vii) Sealant applied with a spray gun should be applied in a solid,
continuous pattern.
(viii) Allow sealant to dry or cure to manufacturer's recommendations.
(ix) When required, prime sealant as soon as it no longer feels tacky,
then topcoat as necessary.
(5) Faying surface sealant is applied between the contacting surface
of two or more parts, and is the most effective seal that can be produced.
It should be used for all assembly and, where possible, reassembly. Where
at all possible it should be used in conjunction with fillet sealing. There
are two types of faying surface seal installations, removable and permanent.
(i) The removable type is for access doors, removable panels, inspection
plates, windows, etc. The sealant is normally applied to the substructure
and a parting agent applied on the removable panel during sealant cure.
(ii) The permanent type is for sealing between parts of a structure
that is permanently fastened together with a high adhesion sealant.
(6) The fillet, or seam, seal is the most common type found on an aircraft.
Fillet seals are used to cover structural joints or seams along stiffeners,
skin butts, walls, spars, and longerons, and to seal around fittings and
fasteners. It should be used in conjunction with faying surface sealing
and in place of it if the assembly sequence restricts the use of faying
surface sealing.
(7) Injection sealing is used primarily to fill voids created by structural
joggles, gaps, and openings. Use only those sealants recommended by the
aircraft/equipment manufacturer. Force sealant into the area using a sealant
gun. This method is a means of producing a continuous seal where it becomes
impossible to lay down a continuous bead of sealant while fillet sealing.
Clean the voids of all dirt, chips, burrs, grease, and oil before injection
sealing.
(8) The fastener sealing method depends on the type of fastener. Fasteners
are sealed either during assembly or after assembly. To seal a permanent
fastener during assembly, apply the sealant to the hole or dip the fastener
into the sealant, and install the fastener while sealant is wet. For removable
fasteners, start the fastener in the hole and apply sealant to the lower
side of the fastener head or countersink. To seal after assembly, apply
sealant to the fastener head after installation.
(9) Sealing of fuel cells should be accomplished per the aircraft manufacturer's
maintenance manual procedures.
g. Paint Finishes and Touchup Procedures.
(1) The primary objective of any paint system is to protect exposed
surfaces against corrosion and other forms of deterioration. Operational
uses for particular paint schemes include:
(i) High visibility requirements;
(ii) Identification markings;
(iii) Abrasion protection; and
(iv) Specialty coatings (i.e., walkway coatings).
(2) The paint system on aircraft consists of a primer coat and a topcoat.
The primer promotes adhesion and contains corrosion inhibitors. The topcoat
provides durability to the paint system, including weather and chemical
resistance, along with the coloring necessary for operational requirements.
(3) Some aircraft surfaces (teflon filled, rain erosion, walkways, etc.)
require specialized coatings to satisfy service exposure and operational
needs. For these surfaces, refer to the specific manufacturer's maintenance
manual for the aircraft in question.
(4) The Environmental Protection Agency, as well as certain local air
pollution control districts, has implementated rules which limit the volatile
organic content (VOC), or solvent content, of paints applied to aircraft
and ground support equipment. It is the responsibility of the user to insure
that these rules are understood and obeyed. FAILURE TO COMPLY WITH CURRENT
RULES CAN RESULT IN LARGE FINES.
(5) Much of the effectiveness of a paint finish and its adherence depend
on careful preparation of the surface prior to touch up and repair.
(i) Aged paint surfaces must be scuff sanded to ensure adhesion of overcoated,
freshly applied paint. Sanding requires a complete roughening of the paint
surface and can be accomplished by hand sanding or with the use of power
tools.
(ii) For final preparation, ensure that surfaces to be painted are free
of corrosion, have been prepared and the surrounding paint feathered, have
been cleaned, and have been conversion coated. Replace any seam sealants
when necessary. Mask areas as required to prevent overspray.
(6) Spray application for touchup, overcoat and total repaint:
(i) Primers should be thinned with the applicable thinner as required
and recommended by the paint manufacturer, stirred, and applied in even
coats. Primer thickness varies for each type primer but generally the total
dry film thickness is 0.6 to 0.9 mils (0.0006 to 0.0009 inch). You should
be able to see through this film thickness. Allow primer to air dry prior
to topcoat application in accordance with the paint manufacturer's recommendations.
Normally topcoat application should occur within 24 hours after primer
application.
(ii) Topcoats should be thinned with the applicable thinner as required
and recommended by the paint manufacturer, stirred, and applied in even
coats. Topcoat thickness varies for each topcoat but generally the total
dry film thickness is 1.5 to 2.0 mils (0.0015 to 0.002 inch). Allow the
topcoat to air dry in accordance with the paint manufacturer's instructions.
(A) Teflon filled (antichafe) coatings should be applied over a primer
in accordance with the manufacturer's instructions.
(B) Walkway compounds should be applied over a primer in accordance
with the manufacturer's instructions.
413 - 499 RESERVED.